Peroral Endoscopic Myotomy (POEM) for Achalasia
Abstract
Achalasia, a primary motility disorder of the esophagus, is the result of improper relaxation of the lower esophageal sphincter and has an incidence ranging from 1 to 6 in 100,000. Impaired transit of food and liquid from the esophagus to the stomach results in symptoms of dysphagia, regurgitation, retrosternal fullness/pain, and weight loss. Symptoms can be managed with a range of medical or procedural therapy. However, the best results are obtained from surgical management with myotomy. Peroral endoscopic myotomy (POEM) has emerged as a less invasive manner through which to perform a myotomy and provides relief of dysphagia comparable to laparoscopic Heller myotomy – the current standard of surgical therapy for achalasia.
Case Overview
Background
Achalasia is a primary motility disorder of the esophagus with an incidence ranging from 1 to 6 in 100,000.1,2 Achalasia is due to inappropriate relaxation of the lower esophageal sphincter (LES), resulting in impaired transit of a food bolus from the esophagus to the stomach. Consequently, peristalsis in the esophagus is impaired with a resultant increase in simultaneous contractions of the esophagus with lower contractile amplitude.2
The pathophysiology of achalasia is believed to be due to degeneration of the ganglia of the esophageal myenteric plexus.2 Hypertension in the LES develops with subsequent dilation of the esophagus and retention of food, liquids, and saliva in the esophagus. As a result, patients typically experience dysphagia, regurgitation, and retrosternal pain or fullness. Many symptoms develop slowly, and the mean time to diagnosis is approximately 4–5 years.1
Focused History
The patient is a 56-year-old female with a past medical history of obesity and type 1 achalasia who presented with two years of dysphagia associated with solids. Over the last six months, her dysphagia had progressed; she was now experiencing dysphagia with liquids. After a meal, she experienced a sensation of retrosternal fullness that was alleviated by emesis. She had no complaints of weight loss or regurgitation.
Of note, the most common presenting symptom of achalasia is dysphagia of solids (experienced by up to 90% of patients) and liquids (85% of patients).1
Physical Exam
The physical exam for achalasia is largely nonspecific with the history of present illness offering greater insight into the potential diagnosis of achalasia and other esophageal motility disorders. Depending on the length of time that the patient has been experiencing symptoms, patients may display signs of weight loss.
In this particular case, the patient’s physical exam was largely unremarkable. She was well-appearing but obese with a body mass index (BMI) of 36. Her abdomen was soft, non-distended, and non-tender.
Imaging
Once the clinical history has raised the index of suspicion for achalasia, several imaging modalities are used to confirm the diagnosis.
Esophagogastroduodenoscopy (EGD) is essential to rule out other causes of mechanical obstruction in the distal esophagus, such as malignancy or peptic stricture, that could produce symptoms of pseudoachalasia. Both of these alternative diagnoses can mimic the symptoms of achalasia but are not managed with myotomy. Ruling out infiltrating malignancies of the esophagus or gastric cardia is of paramount importance in the evaluation of a patient with suspected achalasia. Furthermore, EGD is useful to assess the mucosa for any mucosal inflammatory changes that may affect management.
Esophageal manometry is extremely useful in differentiating achalasia from other esophageal motility disorders. Manometry can be performed as either conventional manometry (CM) with a line tracing or as high-resolution manometry (HRM) with esophageal pressure topography. HRM provides pressure measurements at approximately 1-cm intervals down the esophagus compared to the 3–5-cm intervals of CM. The greater resolution of HRM has allowed clinicians to identify three subtypes of achalasia (based on the Chicago Classification3): 1) Aperistalsis, 2) Pan-esophageal pressurization, 3) Spastic peristalsis.4
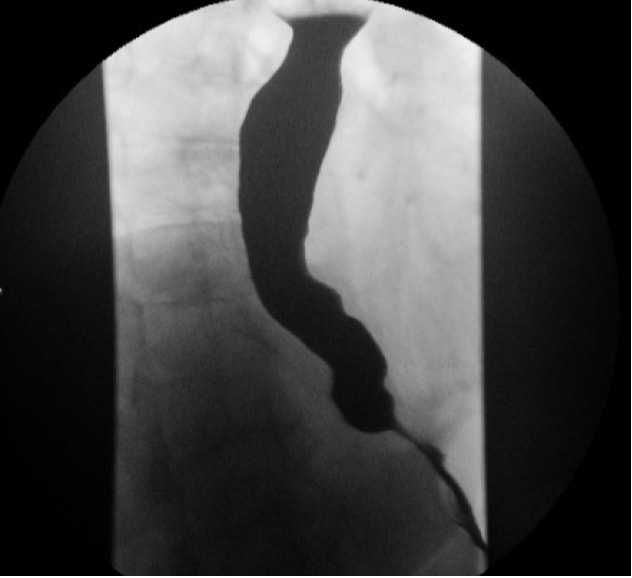
Contrast esophagram (eg, barium swallow) allows for an assessment of anatomic changes to the esophagus that result from achalasia, such as tortuosity, angulation, and degree of dilation. The classic “bird’s beak” appearance of the esophagus (Fig. 1) is frequently seen in achalasia. Timed emptying of barium from the esophagus is a very useful modality of quantifying outflow obstruction both before and after therapeutic intervention.
Natural History
As previously discussed, the underlying pathophysiology of achalasia is due to non-relaxation of the LES. As the disease progresses, dilation of the esophagus is exacerbated; a greater volume of food, liquid, saliva, and air can be retained within the esophagus. As dilation of the esophagus worsens, the esophagus can become tortuous as well. Symptoms progress from initial dysphagia to regurgitation and retrosternal chest pain. Patients with severe disease develop weight loss as a consequence of their dysphagia. Furthermore, irritation of the mucosa from retained particles in the esophagus can lead to mucosal inflammation and may contribute to the 16-fold increased risk of esophageal cancer seen in patients with achalasia.5
Options for Treatment
Endoscopic pneumatic dilation is one of the first options for treatment of achalasia, but it often requires multiple treatments and carries a risk of perforation.
Endoscopic intrasphincteric injection of botulinum toxin has also been demonstrated to have some effect in alleviating symptoms of dysphagia in achalasia. However, effects wane within a year, and recurrent injection or other forms of treatment are necessary to continue symptomatic relief.6 Botulinum therapy is largely reserved for patients who are unable to tolerate surgical management.
Heller myotomy, which involves division of the muscular layers of the esophagus, is associated with better long-term results in the management of achalasia than either pneumatic dilation or botulinum injection.7,8
Peroral endoscopic myotomy (POEM) has been increasingly utilized to treat achalasia. POEM endoscopically divides the LES muscle fibers similar to laparoscopic Heller myotomy, but the route of access is less invasive as no incisions or trocars need to be used.
Rationale for Treatment
The risks and benefits of POEM versus laparoscopic Heller myotomy were discussed with the patient. The patient was diagnosed with Type I achalasia, for which either POEM or Heller myotomy are expected to have similar outcomes. Given the patient was relatively young and healthy with no evidence of mucosal inflammation or disease on his upper endoscopy, the patient was expected to have better results from surgical management of his achalasia than with pneumatic dilation.
Special Considerations
Contraindications to performance of POEM include a prior history of esophageal surgery, portal hypertension, and coagulopathy.
Discussion
Natural Orifice Transluminal Endoscopic Surgery (NOTES) developed as minimally invasive surgeons and endoscopists sought to find less invasive methods for surgical management of disease. Although several NOTES procedures and routes have shown to be safe, feasible, and reproducible in clinical practice, POEM is perhaps the most widely performed NOTES procedure at this time after years of development and research on its safety as well as its short- and long-term effectiveness.9
POEM was first developed in Japan in 200810 (and later introduced in the US in 20109) as an alternative to laparoscopic Heller myotomy (LHM) for the management of achalasia. While an adequately powered randomized controlled trial has yet to be published comparing the outcomes of POEM with LHM, prospective observational studies have established that dysphagia relief rates after POEM range from 83% to 89% with remission of symptoms at one year.11 More objectively, POEM has resulted in a reduction of mean LES pressure by 62%.12,13 As with open and laparoscopic myotomy, the key to successful surgical treatment of achalasia with POEM is to completely divide all circular muscle fibers of the esophagus to allow for relaxation of the LES.
Preoperatively, clinicians may calculate an Eckardt score, a qualitative survey that assesses the severity of symptoms based on degree of dysphagia, symptom duration, and frequency of symptoms such as regurgitation.14 Such scores can be utilized to standardize comparison of symptoms before and after POEM,12,13 and some studies have demonstrated up to an 87% reduction in scores after POEM.12
Nonsurgical approaches to achalasia have varying rates of success with variable risk profiles for complications. The least effective is medical therapy alone. Drugs such as calcium channel blockers and long-acting nitrates function by facilitating smooth muscle relaxation to lower LES pressure. However, the effects are short acting (ranging from 30 to 120 minutes) and offset by pharmacological side effects such as hypotension or headache.15 Endoscopic pneumatic dilation requires repeated, graded intervention to achieve symptom relief rates approaching that of LHM. Furthermore, dilation is associated with a 1.9% risk of esophageal perforation even when it is performed by experienced endoscopists.16
A recent systematic review and meta-analysis comparing LHM and POEM found that relief of dysphagia at 12 months was better in patients who underwent POEM (93.5% relief) versus LHM (91%) and that the difference persisted at 24 months (POEM 92.7% vs. LHM 90%).17 Patients who underwent POEM were more likely to develop symptoms of gastrointestinal reflux disease (GERD) as well as endoscopic findings of GERD such as esophagitis (POEM 47.5% vs. LHM 11.1%), but all of the LHM patients included in this study had also undergone a fundoplication.17 In patients who undergo POEM and develop GERD, a laparoscopic fundoplication can be performed if symptoms are not controlled with pharmacologic therapy alone.
At our institution, we obtain a contrast esophagogram on postoperative day one to rule out leaks and to assess the passage of contrast from the esophagus into the stomach. We also maintain patients on a liquid diet for the first week after surgery given that postoperative edema may affect the passage of solid foods. After one week, patients are cleared to resume a regular diet with solid foods. Routine follow-up is scheduled for two weeks, three to six months, and one year after surgery. We routinely prescribe acid suppression therapy with proton pump inhibitors (PPI) to postoperative patients for a minimum of one year. If patients wish to stop their PPI at one year, we perform a pH study to assess for GERD.
This patient’s postoperative esophagogram was unremarkable, and the patient was discharged on a liquid diet for one week. She progressed without issue to a solid diet. Her postoperative follow-up has been unremarkable, and she continues to do well.
Equipment
We used a forward view HD endoscope, TT knife KD-640L (Olympus, Tokyo, Japan), Transparent Cap MH-588 (Olympus, Tokyo, Japan), and VIO 300D ERBE generator (Tubingen, Germany).
Disclosures
Drs. Meireles and Rattner are consultants for Olympus. Drs. Meireles and Hashimoto have received grant funding from the Natural Orifice Consortium for Assessment and Research (NOSCAR). Dr. Hashimoto is partially funded by the MGH Edward D. Churchill Surgical Education and Simulation Research Fellowship and the National Institute of Diabetes and Digestive and Kidney Diseases (Grant #: T32DK007754-17).
Statement of Consent
The patient referred to in this video article has given his informed consent to be filmed and is aware that information and images will be published online.
Citations
- Burakoff R, Chan WW. Overview of esophageal motility disorders. In: Sugarbaker DJ, Bueno R, Colson YL, Jaklitsch M, Krasna MJ, Mentzer S, eds. Adult Chest Surgery. 2nd ed. New York, NY: McGraw-Hill Education; 2015.
- Jobe BA, Hunter JG, Watson DI. Esophagus and diaphragmatic hernia. In: Brunicardi FC, Andersen DK, Billiar TR, et al, eds. Schwartz's Principles of Surgery. 10th ed. New York, NY: McGraw-Hill Education; 2014.
- Bredenoord AJ, Fox M, Kahrilas PJ, Pandolfino JE, Schweizer W, Smout AJ; International High Resolution Manometry Working Group. Chicago classification criteria of esophageal motility disorders defined in high resolution esophageal pressure topography. Neurogastroenterol Motil. 2012;24(suppl 1):57-65. doi:10.1111/j.1365-2982.2011.01834.x.
- Rohof WO, Salvador R, Annese V, et al. Outcomes of treatment for achalasia depend on manometric subtype. Gastroenterology. 2013;144(4):718-725. doi:10.1053/j.gastro.2012.12.027.
- Sandler RS, Nyrén O, Ekbom A, Eisen GM, Yuen J, Josefsson S. The risk of esophageal cancer in patients with achalasia: a population-based study. JAMA. 1995;274(17):1359-1362. doi:10.1001/jama.1995.03530170039029.
- Pasricha PJ, Ravich WJ, Hendrix TR, Sostre S, Jones B, Kalloo AN. Intrasphincteric botulinum toxin for the treatment of achalasia. N Engl J Med. 1995;332(12):774-778. doi:10.1056/NEJM199503233321203.
- Zaninotto G, Annese V, Costantini M, et al. Randomized controlled trial of botulinum toxin versus laparoscopic Heller myotomy for esophageal achalasia. Ann Surg. 2004;239(3):364-370. doi:10.1097/01.sla.0000114217.52941.c5.
- Urbach DR, Hansen PD, Khajanchee YS, Swanstrom LL. A decision analysis of the optimal initial approach to achalasia: laparoscopic Heller myotomy with partial fundoplication, thoracoscopic Heller myotomy, pneumatic dilatation, or botulinum toxin injection. J Gastrointest Surg. 2001;5(2):192-205. doi:10.1016/S1091-255X(01)80033-0.
- Horgan S, Meireles OR, Jacobsen GR, et al. Broad clinical utilization of NOTES: is it safe? Surg Endosc. 2013;27(6):1872-1880. doi:10.1007/s00464-012-2736-z.
- Inoue H, Minami H, Satodate H, Kudo SE. First clinical experience of submucosal endoscopic esophageal myotomy for esophageal achalasia with no skin incision. Gastrointest Endosc. 2009;69(5):AB122. doi:10.1016/j.gie.2009.03.133.
- Von Renteln D, Fuchs KH, Fockens P, et al. Peroral endoscopic myotomy for the treatment of achalasia: an international prospective multicenter study. Gastroenterology. 2013;145(2):309-311.e3. doi:10.1053/j.gastro.2013.04.057.
- Inoue H, Minami H, Kobayashi Y, et al. Peroral endoscopic myotomy (POEM) for esophageal achalasia. Endoscopy. 2010;42(4):265-271. doi:10.1055/s-0029-1244080.
- Swanstrom LL, Kurian A, Dunst CM, Sharata A, Bhayani N, Rieder E. Long-term outcomes of an endoscopic myotomy for achalasia: the POEM procedure. Ann Surg. 2012;256(4):659-667. doi:10.1097/SLA.0b013e31826b5212.
- Eckardt AJ, Eckardt VF. Treatment and surveillance strategies in achalasia: an update. Nat Rev Gastroenterol Hepatol. 2011;8(6):311-319. doi:10.1038/nrgastro.2011.68.
- Vaezi MF, Richter JE. Current therapies for achalasia: comparison and efficacy. J Clin Gastroenterol. 1998;27(1):21-35. doi:10.1097/00004836-199807000-00006.
- Vaezi MF, Pandolfino JE, Vela MF. ACG clinical guideline: diagnosis and management of achalasia. Am J Gastroenterol. 2013;108(8):1238-1249. doi:10.1038/ajg.2013.196.
- Schlottmann F, Luckett DJ, Fine J, Shaheen NJ, Patti MG. Laparoscopic Heller myotomy versus peroral endoscopic myotomy (POEM) for achalasia: a systematic review and meta-analysis. Ann Surg. 2018;267(3):451-460. doi:10.1097/SLA.0000000000002311.