Leiomyosarcoma of Inferior Vena Cava: Resection and Reconstruction
Abstract
Primary leiomyosarcomas of the inferior vena cava (IVC) are rare tumors with complex anatomical relationships. Surgical resection remains the primary approach for management, with selective use of preoperative radiation and chemotherapy. Given the propensity for local invasion of these tumors, radical resection of surrounding structures is often required. Herein we describe the presentation, work-up, operative management, and outcomes of these lesions through the case of a patient with a tumor involving the middle segment of the IVC. Given the extent of involvement, IVC resection with en bloc right nephrectomy, right adrenalectomy, and partial left renal vein resection was performed with vascular reconstruction using a prosthetic graft. With appropriate preoperative planning and a well-coordinated multidisciplinary approach, aggressive surgical resection can be safely performed and patients can benefit from favorable long-term survival.
Case Overview
Background
Representing less than 0.5% of all soft tissue sarcomas, leiomyosarcoma (LMS) of the inferior vena cava (IVC) is an uncommon neoplasm involving the smooth muscle of the vein.1 The disease is more commonly seen in females (3:1) in the fifth decade of life.2 Maximum tumor diameter has been noted to range between 8–37 centimeters.3 In addition to involvement of the right kidney or adrenal, direct extension to the right or left liver, infrarenal aorta, intracardiac region, and supradiaphragmatic right lung have been noted.3 In the majority of cases, patients receive a preoperative histologic diagnosis by either percutaneous biopsy or laparotomy.3
Focused History of Patient
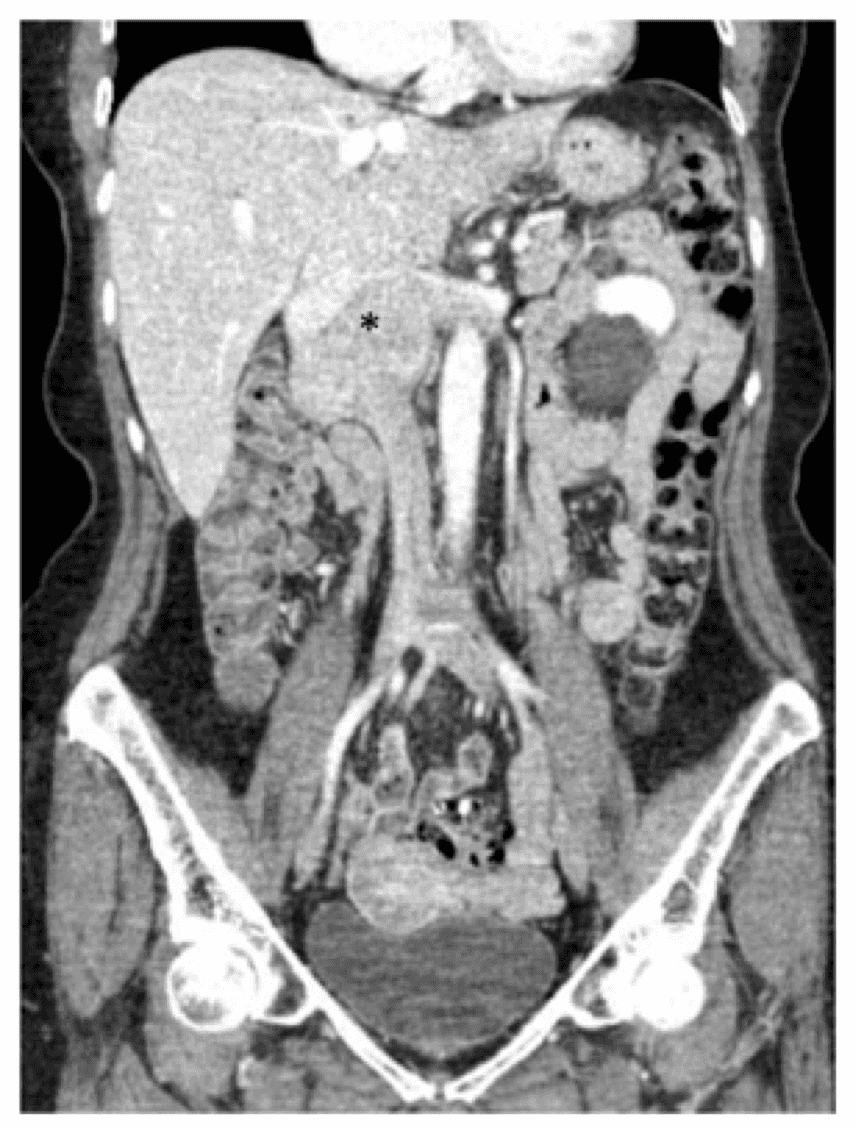
A 73-year-old female presented with a history of intermittent lower back pain, lower extremity swelling, and a six-pound weight loss over the course of a year. Her past medical history was notable for hypertension for which she took atenolol, hydrochlorothiazide, and nifedipine as well as reflux for which she was maintained on omeprazole. Upon workup by her primary care physician, she received a computed tomography (CT) scan of the abdomen and pelvis demonstrating a mass along the juxtarenal IVC that was biopsied and found to be consistent with leiomyosarcoma (Figure 1).
Physical Exam
The most common presenting symptom for patients with IVC LMS is abdominal pain (59.9%), followed by lower extremity edema (14.8%), weight loss (11.4%), back pain (9.7%), and abdominal distension (9.3%).1-4 Lower extremity edema is thought to only occur in a fraction of patients likely due to the development of venous collaterals in the setting of slow tumor growth. Much less frequently, deep venous thrombosis (1.7%) or Budd-Chiari syndrome (1.3%) are responsible for presentation.1-4 Additional signs such as cardiac arrhythmia may be present in patients with intracardiac tumor extension into the right atrium. As mentioned, our patient presented with a history of intermittent lower back pain, lower extremity swelling, and weight loss. On physical exam, however, she was well-appearing, had a soft, non-tender abdomen without hepatosplenomegaly, and was noted to have non-edematous distal extremities. Her exam was otherwise unremarkable.
Imaging
Axial imaging using CT or magnetic resonance imaging (MRI) constitutes the primary imaging modality for evaluating patients presenting with LMS as these scans provide details on the extent of tumor, local invasion, and distant disease. On CT, LMS of the IVC appears as a heterogeneous, non-calcified mass with peripheral enhancement.5 MRI images are notable for low signal intensity on T1 weighted images and high signal intensity on T2 weighted images.5 A portion of patients may also receive abdominal ultrasound, where real-time Doppler may delineate the degree of vascular obstruction and thrombosis. Although invasive studies such as ascending or retrograde cavography and selective arteriography have been advocated by some authors,3 they are becoming less common given the increased resolution and reconstructions that can be obtained through noninvasive axial imaging. CT images may be further enhanced by delayed phase or venous phase images that can better delineate the IVC and residual flow lumen. Lastly, in patients in which there is concern for intracardiac extension of tumor, transesophageal echocardiography should be obtained preoperatively.
The patient in this report underwent a CT scan with contrast of the abdomen and pelvis, and we noted a 7.5-cm mass involving the IVC beginning at the level of the renal veins (the right renal vein was extensively involved and there was also involvement of the origin of the left renal vein) to the level of—but not involving—the hepatic veins (Figure 1). The duodenum and pancreas were noted to be displaced anteriorly without evidence of direct invasion.
Natural History
Primary IVC LMS has been found to be uniformly fatal and without 3-year survival in patients who received incomplete resection.1 On pooled analysis, in patients undergoing surgical resection, 1- and 5-year disease-free survival (DFS) has been noted to be low (57% and 6%, respectively). However, overall survival has been favorable (92% and 55%, respectively).2
Options for Treatment
Complete surgical resection is the preferred treatment for patients who can tolerate surgery and often necessitates en bloc resection of involved adjacent organs. Resection with ligation of the IVC can be performed for infrarenal tumors or those necessitating resection of the right kidney in which substantial collaterals for the left kidney have formed. In approximately a quarter of patients, partial IVC resection can be performed.3 It has been suggested that if < 75% of the IVC is resected, primary resection or patch cavoplasty can be used, but in cases where > 75% of the circumference is removed, complete resection and reconstruction is needed.6 For those requiring reconstruction, a ringed polytetrafluoroethylene (PTFE) prosthesis can be sized to match and can be sutured in place with a 4-0 or 5-0 polypropylene suture. The use of chemotherapy or radiotherapy either as neoadjuvant or adjuvant therapy may have an increasing role. However given the rarity of this disease and the paucity of treatment outcome data available to date, these modalities are currently tailored to each individual patient after multidisciplinary discussion, and there is variability in their application.
Rationale for Treatment
Given the rarity of primary IVC LMS, there are limited studies supporting a non-operative approach, and complete surgical resection remains the optimal management strategy.2 In efforts to decrease the risk of recurrence at the retroperitoneal margin as well as tumor seeding of the peritoneal cavity at the time of surgical resection, our institutional practice for the management of retroperitoneal sarcoma involves neoadjuvant radiation therapy followed by radical resection and chemotherapy if deemed necessary after reviewing and discussing final explant pathology. Specifically, our patient was enrolled in a National Cancer Institute (NCI) sponsored trial (DFCI/HCC 12-100) in which they were treated with 63 Gray (Gy) to the retroperitoneal margin and 50.4 Gy to the whole tumor area using pencil beam scanning protons over five weeks. The patient tolerated this well, had a stable re-staging scan, and subsequently proceeded with radical resection of her retroperitoneal sarcoma with en bloc resection of the right kidney, the right adrenal gland, the inferior vena cava, and a portion of the left renal vein as detailed below (see procedure outline).
Discussion
The first description of a primary IVC LMS was in 1871 by Perl,7 followed by the first resection by Melchior in 1928 in which the patient died 2 weeks postoperatively.8 With advancements allowing for preoperative planning, refinement of surgical technique and exposure, and improvements in critical care, 30-day perioperative mortality after resection of IVC LMS is now 1.9%.2 However, there remains substantial 30-day morbidity (24.7%).2 Nonetheless, surgical resection remains the primary treatment strategy. With this approach, long term 5-year DFS has been noted to be low (6%), whereas 5-year overall survival has been favorable (55%).2
Until recently, a number of reports in the literature studying IVC LMS were limited by small sample sizes. A pooled analysis by Wachtel et al. has allowed for the most comprehensive study including 377 patients.2 Upon adjusted analysis, factors identified as leading to decreased overall survival included large tumor size (≥9 cm), older age (≥55 years), the need for en bloc resection, positive margin status, adjuvant chemotherapy, and tumor location. Specifically, primary IVC leiomyosarcomas are classified according to extent of venous involvement and are described by the upper limit of the tumor and the degree of tumor extension. Using this classification system, segment I tumors are infrarenal, segment II are interrenal and/or suprarenal up to but not including the suprahepatic veins, and segment III are those that extend into the suprahepatic veins and have possible intracardiac extension. Segment II, or middle segment, tumors are the most common and have been found to have better outcomes than those arising in either segment I or segment III.2,9 In part, this is thought to be due to early recognition of tumors in Segment II due to a closer anatomic proximity of these tumors to multiple organs which may lead to pain or obstruction.2 Our patient’s tumor could best be classified as a segment II lesion.
A few comments regarding the operative procedure for IVC LMS resection and reconstruction warrant further discussion. Regarding exposure, an abdominal approach (median laparotomy or right subcostal incision) has been used in the majority of cases (60%), whereas a thoracoabdominal approach with possible sternotomy can be employed depending on the distinct anatomy of the tumor being resected.3 Specifically, for tumors extending into the retrohepatic or suprahepatic vena cava, total hepatic exclusion by means of clamping the IVC above and below the liver in addition to clamping of the portal vein may be necessary. In patients with intracardiac extension, adequate exposure to allow for cardiopulmonary bypass may be warranted. In general, patients with tumors isolated to the infrahepatic and pararenal cava will likely tolerate complete cross-clamping of the IVC without significant hemodynamic alterations as long as adequate preload is ensured with appropriate intraoperative resuscitation by the anesthesia team. With regards to technique, in patients with venous hypertension (proximal venous pressures of 30 mmHg or greater), reconstruction (as opposed to ligation, primary resection, or cavoplasty) is recommended to prevent postoperative lower extremity edema.3 Furthermore, reimplantation of the renal or hepatic veins may be necessary depending upon the extent of tumor resected. There currently is no standard for postoperative anticoagulation management, and antiplatelet aspirin therapy, as was used in our patient who was discharged on 325 mg of aspirin daily, is associated with low rates of postoperative graft thrombosis.10
Randomized controlled trials evaluating the efficacy of chemotherapy or radiotherapy in the management of IVC LMS may never be published due to the rarity of disease. Final pathology of our patient was notable for a grade 2/3 leiomyosarcoma (yPT2bN0) with negative margins. Upon discussion with medical oncology, we decided she did not require any additional adjuvant therapy. Regarding follow-up, interval CT scans should be used to evaluate local recurrence and distant metastasis based on risk for recurrence.6 We obtained our first surveillance scan at 3 months, and it was normal. In cases of distant recurrence, surgical resection is recommended as is the use of systemic therapy for those unable to tolerate an operation.4
Prolonged survival in patients with IVC LMS is possible and requires a multidisciplinary approach. Although surgery remains the cornerstone of treatment, the involvement of medical and radiation oncology as well as multiple surgical subspecialties (eg, surgical oncology and transplant surgery) is encouraged to optimally manage and individualize treatment for patients with this rare malignancy.
Disclosures
The authors have no relevant disclosures.
Statement of Consent
The patient referred to in this video article has given their informed consent to be filmed and is aware that information and images will be published online.
Citations
- Hollenbeck ST, Grobmyer SR, Kent KC, Brennan MF. Surgical treatment and outcomes of patients with primary inferior vena cava leiomyosarcoma. J Am Coll Surg. 2003;197(4):575-579. doi:10.1016/S1072-7515(03)00433-2.
- Wachtel H, Gupta M, Bartlett EK, et al. Outcomes after resection of leiomyosarcomas of the inferior vena cava: a pooled data analysis of 377 cases. Surg Oncol. 2015;24(1):21-27. doi:10.1016/j.suronc.2014.10.007.
- Kieffer E, Alaoui M, Piette JC, Cacoub P, Chiche L. Leiomyosarcoma of the inferior vena cava: experience in 22 cases. Ann Surg. 2006;244(2):289-295. doi:10.1097/01.sla.0000229964.71743.db.
- Cananzi FCM, Mussi C, Bordoni MG, et al. Role of surgery in the multimodal treatment of primary and recurrent leiomyosarcoma of the inferior vena cava. J Surg Oncol. 2016;114(1):44-49. doi:10.1002/jso.24244.
- Ganeshalingam S, Rajeswaran G, Jones RL, Thway K, Moskovic E. Leiomyosarcomas of the inferior vena cava: diagnostic features on cross-sectional imaging. Clin Radiol. 2011;66(1):50-56. doi:10.1016/j.crad.2010.08.004.
- Dull BZ, Smith B, Tefera G, Weber S. Surgical management of retroperitoneal leiomyosarcoma arising from the inferior vena cava. J Gastrointest Surg. 2013;17(12):2166-2171. doi:10.1007/s11605-013-2385-0.
- Perl L, Virchow R. Ein Fall von Sarkom der Vena cava inferior. Archiv für pathologische Anatomie und Physiologie und für klinische Medicin. 1871;53(4):378-383. doi:10.1007/BF01957198.
- Melchior E. Sarkom der Vena cava inferior. Deutsche Zeitschrift für Chirurgie. 1928;213(1-2):135-140. doi:10.1007/BF02796714.
- Mingoli A, Cavallaro A, Sapienza P, Di Marzo L, Feldhaus RJ, Cavallari N. International registry of inferior vena cava leiomyosarcoma: analysis of a world series on 218 patients. Anticancer Res. 1996;16(5B):3201-3205.
- Fiore M, Colombo C, Locati P, et al. Surgical technique, morbidity, and outcome of primary retroperitoneal sarcoma involving inferior vena cava. Ann Surg Oncol. 2012;19(2):511-518. doi:10.1245/s10434-011-1954-2.